Issue:November/December 2015
DRUG DEVICE MARKET - Joining Forces: Global Markets for Drug-Device Combinations
INTRODUCTION
Products that combine a drug or biologic with a device, such as drug-eluting stents and drug delivery systems, can offer valuable approaches for treating disease. As the purpose of most drug-device combination products is to either augment a device’s efficacy and/or safety through the use of a drug coating, or to use a device to deliver a drug locally and in that way increase efficacy of treatment, these combination products are generally used to treat conditions rather than cure them.
However, some do affect a cure, and the possibility of more curative devices brought to market cannot be discounted as the relevant sciences advance. Consequently, the development of the market for combination products is closely related to the drug delivery systems sector, which represents a vast area of research and the demand for sophisticated drug delivery devices behind many novel product developments. Advanced drug delivery devices offer increased efficiency, improved performance, and convenience.
It is anticipated that combination products will enable the use of therapy candidates that cannot currently be used alone due to systemic effects and toxicities. Combination product technology will enable safer and more effective technologies due to careful and precise drug targeting, local administration, and individualized therapy. These technologies have paved the way for combination products that will help patients suffering from cancer, heart disease, multiple sclerosis, cerebral palsy, spinal-cord injuries, anemia, hepatitis, rheumatoid arthritis, diabetes, and other serious diseases and conditions.
The number of product categories and individual product offerings in the drug-device combination market has grown to be relatively large. Although a couple of categories (drug-eluting stents and antimicrobial catheters) together account for the majority of sales, numerous developmental companies are pursuing products that almost undoubtedly will significantly affect efficacy, outcomes, and economics in medical procedures in the years to come.
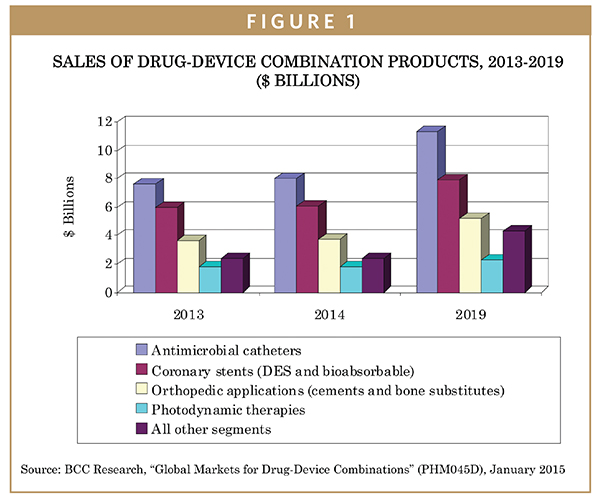
According to BCC Research, sales of drug-device combination products reached $21.4 billion in 2013 and $22 billion in 2014. This market is expected to grow to $31 billion in 2019, with a compound annual growth rate (CAGR) of 7.1% from 2014 to 2019.
MARKET ANALYSIS
The combination products industry includes makers of drug delivery systems, gene therapy systems, personalized medicine drug-device combinations, biological-device combinations, nanotechnology, and certain other products for diagnostic and therapeutic treatments of cardiovascular, metabolic, oncologic, and other disorders.
Overall, the combination product industry is growing relatively rapidly, with particular dynamism in successful niche products, and attracting a lot of startup capital for R&D and developmental companies. Established products, such as drug-eluting stents, anti-microbial catheters, and photodynamic therapy, represent fairly large markets. But going forward, the fastest growth will probably be in even newer areas involving nanotechnology-enabled products.
It is anticipated that combination products will enable the use of therapy candidates that cannot currently be used alone due to systemic effects and toxicities. Combination product technology will enable safer and more effective technologies due to careful and precise drug targeting, local administration, and individualized therapy. These and other drivers have paved the way for combination products that will help patients suffering from cancer, heart disease, multiple sclerosis, cerebral palsy, spinal-cord injuries, anemia, hepatitis, rheumatoid arthritis, diabetes, and other serious diseases and conditions.
The R&D effort and accompanying expenditures in combination products is very substantial. A large segment of the market is drug-eluting stents. The DES stent market historically was growing at approximately 8% per year. That growth trend is projected to slow, however, to a CAGR of 5.3% due to commoditization of the stent market and concerns about over-use. Thus, the market for drug-eluting stents is projected to increase from $6 billion in 2013 to $7.9 billion in 2019.
Demand for bone graft substitutes and antibiotic bone cements is growing at a healthy 7% CAGR. Many of the newer niche categories, such as ocular products, drug-eluting beads, and many types of nano-based products, will show significant growth in the range of 8% to 10% per year. Aggregated, these newer niche segments are growing at about 12% per year.
TECHNOLOGY & PATENT ACTIVITY
The drug-device combination industry is a prototypical example of technological innovation as reflected in the patent database. In fact, if only two words were allowed to describe the salient characteristic of the overall industry it would be “technological dynamism.” Indeed, the number and technological import of patents relating to products in this industry reflect the enormous efforts and expenditures to develop breakthrough therapies for serious human diseases.
Large numbers of patents are being issued, almost on a weekly basis. Technology developments are key forces in the industry. Some examples of this are in the area of nanotechnology-enabled devices and small particles, medical device coatings, and delivery systems. All of this activity is driven by the fact that from the standpoint of drug-device combination developers, there is large clinical potential as well as substantial investment returns.
Throughout the past 5 years, each year, about 300 original premarket applications for combination products have been received at the FDA’s Office of Combination Products. According to the latest data, the FDA received 266 original premarket applications for combination products in 2012, 285 applications in 2011, and 311 in 2010. The FDA obviously is a central focus in this industry. Comparable regulatory entities exist overseas. In fact, as the combination product industry has evolved, the FDA’s oversight of this particular segment has become ever more focused.
Because combination products involve components that would normally be regulated under different types of regulatory authorities, and frequently by different FDA Centers, they also raise challenging regulatory, policy, and review management issues. The differences in regulatory pathways for regulatory processes of all aspects of the product life cycle, including preclinical testing, clinical investigation, marketing applications, manufacturing and quality control, adverse event reporting, promotion and advertising, and post-approval modifications.
HIGHLY ADVANCED TECHNOLOGIES AS A MARKET CORE
The pharmaceutical industry spends more on R&D, relative to its sales revenue, than almost any other industry in the US. According to various estimates, the industry’s real (inflation-adjusted) spending on drug R&D has grown between three-fold and six-fold throughout the past 25 years, and that increase has been closely matched by growth in drug sales.
The range of R&D re-investment as a percentage of sales is anywhere from 2% to 25%, depending on the company. Total world pharmaceutical and healthcare R&D is approximately $100 billion per year.
Despite those increases, there has been little change in the number of innovative new drugs approved for use each year, even though the federal government has streamlined its drug approval process. And, only about one-third of the drugs approved annually in the US are new compounds; the rest represent modified forms of, or new uses for, existing drugs.
However, combination products represent an area of growing potential as more and more incorporate cutting-edge, novel technologies that hold great promise for advancing patient care. Beyond drug-eluting stents and inhaled insulin, breakthrough new products approved after the FDA’s OCP was established include drug delivery systems, pharmacogenomic drug-device combinations, nanotechnology, gene therapy systems, and products for many other diagnostic and therapeutic treatments.
Firms develop new drug products in response to various factors. These include likely demand in a given drug market, influenced by available health insurance coverage; doctors’ prescribing practices; demographic changes; government policy toward drug safety and innovation; and the pace of scientific advances in the understanding and treatment of disease. A foundation of combination products is that they typically rely on very sophisticated scientific technologies, such as nanotechnology, genomics, molecular diagnostics, tissue engineering, and stem cell research. In addition to these technologies, the mere convergence of regulated articles fosters novel approaches to treatment and diagnosis: the combinations allow the best of all worlds to confront today’s health problems.
NANOTECHNOLOGY & COMBINATION PRODUCTS
Nanotechnology applications in drug delivery constitute about half of the applications of nanotechnology in medicine. In vitro and in vivo diagnostics and implant technology are the remaining successful realms. There is a large and growing number of organizations involved in drug-device or combination product applications that involve nanotechnology concepts.
Examples of companies involved include Cerulean Pharma, which has a liposomal nanopharmaceutical, CRLX101, currently in development. Immune Pharmaceuticals (Cambridge, MA) is evaluating the use of its NanomAb platform, a second-generation antibody drug conjugate technology, along with chemotherapeutics.
Celgene markets the nanotech combination drug Abraxane. In May 2014, AADi LLC, a clinical-stage biopharmaceutical company focused on treating diseases uniquely suited for nanotechnology approaches, licensed ABI-009 from Celgene. ABI-009 is the nanoparticle albumin-bound version of the mTOR inhibitor sirolimus or rapamycin and leverages the same technology behind Abraxane. Abraxane is a protein-bound, injectable formulation of paclitaxel, a mitotic inhibitor drug used in the treatment of breast cancer. In this formulation, paclitaxel is bonded to albumin as a delivery vehicle.
The FDA approved Abraxane in 2005, and the European Medicines Agency approved it in 2008 for breast cancer, in which cancer did not respond to other chemotherapy. A Phase III trial reported in 2010 showed positive results in first-line non-small-cell lung cancer (NSCLC) when compared with Taxol. However, in 2012 at the American Society of Clinical Oncology meeting, researchers reported that Abraxane did not extend life compared to traditional, and much lower cost, treatment regimes. Total revenue from the sales of Abraxane is presently about $350 million per year. Celgene has said it expects revenues to exceed $1 billion annually if Abraxane is used for other types of cancerous tumors.
As of mid-2014, at least 150 nanotech-based drugs and delivery systems and an additional 140 devices or diagnostic tests were in preclinical, clinical, or commercial development. There are at least 50 actually vended drugs or devices that are wholly nanotechnology based according to Nature Biotechnology. The National Science Foundation has predicted that nanotechnology will produce half of the pharmaceutical industry product line by 2015.
World demand for nanomedicines, not all of which are combination product based, is forecast to increase annually by about 12% to 15%, reaching at least $95 billion by 2019. Therapeutic monoclonal antibodies are expected to comprise a significant portion of the nanomedicine market, as more than a third of biotechnology development projects are seeking to apply those proteins to treat a wide array of conditions. That sector alone was estimated to generate $31 billion in 2012. Other major nanomedicine market segments include polymer-based drugs and crystalline nanomedicines. The global nanomedicine market for the central nervous system (CNS) products market is expected to grow to $29.5 billion by 2016. The anticancer products market is expected to reach $12.7 billion by 2016.
At present, there are more than 22,000 scientific publications and 1,500 patent applications per year related to nanotechnology. The exponential increase in scientific publications and patents is the result of increased discovery and investment in nanotechnology that will likely result in substantial and continual changes in products falling under the regulatory authority of the FDA. The FDA has acknowledged that these increases in the discovery, research, and marketing of combination products will have a significant impact.
This article is based on the following market analysis report published by BCC Research: Global Markets for Drug-Device Combinations (PHM045D) by Kevin Gainer.
To view this issue and all back issues online, please visit www.drug-dev.com.
Kevin James Kinsella is a New York City-based healthcare and medical communications professional with more than 15 years of experience in the private and public health sectors.
Kevin Gainer holds both BA and MA degrees in Quantitative Economic Analysis and Forecasting, and has 26 years of economic forecasting, industry intelligence, and market research experience.
Total Page Views: 3851