Issue:March 2015
MARKET BRIEF - Miniaturizing Healthcare - From Microelectronics to Nanobiosensing
INTRODUCTION
Technology miniaturization has challenged scientists for decades. Starting with microelectronics, the concept of miniaturization has been studied and applied to life sciences and biotechnology with prospective results. Molecular behavior at micro- and even nanoscale has demonstrated to be substantially different than at the bulk scale. Under this scope, micro- and nanofluidics technology allows researchers to achieve a more profound understanding of these variations aiming to optimally exploit their benefit in a broad spectrum of application fields.
The principal impact of this technology keeps highest levels for high-throughput screening (HTS) methods for DNA sequencing, in which nanobiochips are mainly used to stretch DNA strands. Similarly, nanofluidic devices are strategically targeted to be used as disposable devices, specially applied to point-of-care (POC) diagnostics.
In other words, as the information revolution has constituted the predominant event in the economy of the 20th century with the silicon microchip or semiconductor, the past decade has evidenced the simultaneous advances in miniaturizing microchip elements on one hand and self-assemble nanoparticles on the other. In fact, performed with the aim of reducing costs while enhancing performance, these approaches have derived in the acceleration of nanotechnology to biochips from an archetypal synthesis of top-down and bottom-up developments, constituting the key to the success of microchip technology.
This approach has also revolutionized the technology transfer model from academia and research organizations to industry, strengthening the conjunct efforts among governmental, industry, and university actors.
Life sciences research and biomedical engineering devices constitute the most significant application targets. Experts’ opinions agree that in set-up, the major impact of nanofluidics technology, including a fluent commercialization and a wide scale adoption, from 2016, is a continuous and prospective advance toward novel fields of application, involving a synergistic convergence of technology clusters. Indeed, the proliferation of lower cost microfluidics-based genomics tools offering improved capabilities and allowing more access to end-users is expected to drive this technology for pharmaceutical and biomedical research throughout the next 5 years.
Working with micro- and nanoliter quantities of reagents in micro- and nanoscale dimensions, these novel devices allow sample separation and detection with greater sensitivity and specificity. The economic burden of carrying laboratory-scale experiments utilizing expensive equipment is significantly reduced, hence obtaining rapid and reliable results. Moreover, the short footprint of these pieces enables fabrication of portable diagnostic devices, importantly enhancing the access of different end-users to medical diagnosis. This fact possesses remarkable importance in developing nations where expensive medical diagnostic facilities are not available to all.
Another remarkable advantage relies on their multiplexing capabilities. Micro- and nanofluidic devices can be used to process multiple samples in parallel, becoming the process substantially more cost and time effective.
IMPACTING PHARMA, BIOMEDICAL & BIOTECH
Micro- and nanofluidics offer a broad spectrum of novel and potentially valuable developments in several markets, motivating the entrance of new players. Indeed, this technology addresses both the enhancement of existing markets and the creation of new markets. Regarding existing markets, this technology is expected to play a major role in the pharmaceutical industry, singularly optimizing current processes and offering completely new decision supports. New markets, on the other hand, are entering the life sciences research technology cluster, revolutionizing the advance of closely related disciplines.
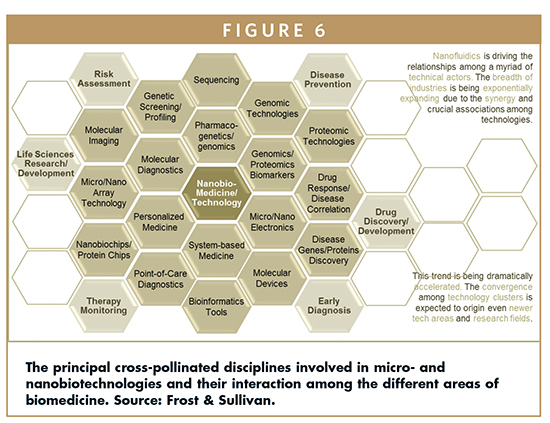
At this point, the remarkable crosspollination from other disciplines and sectors, including the electronics, material sciences, and semiconductor industry into the life sciences and biotech arenas, are surprisingly driving the wide field of nanobiotechnologies.
Special impact is observed on the design of novel biochips for HTS and diagnostics applications, on one hand, and lab-on-a-chip (LOC) and POC devices on the other hand.
CORE TECHNOLOGY APPLICATIONS
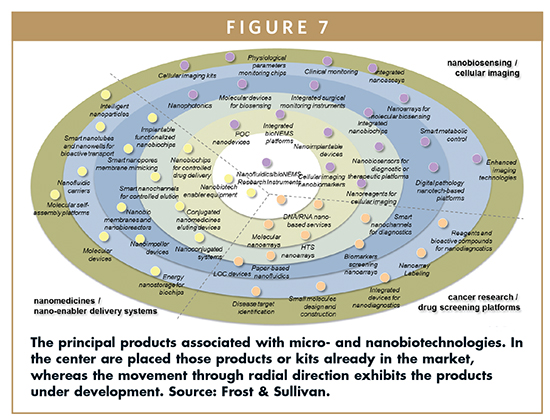
Nanomedicines/Nanoenabler Delivery Systems
• Molecular nanotechnology (MNT) platforms constitute promising tools due to their ability to conform structures and devices at atomic scale precision and accuracy.
• A more specific target delivery through nanoenabler delivery systems allows using lower doses, avoiding cytotoxicity and minimizing side effects, while improving therapy.
Cancer Research/Drug Screening Platforms
• Magnetic or antibody-conjugated targeting can guide nanoparticles to a specific body region, and even cells, aiming to improve cancer therapeutics. Nanoparticles are linked to tumor cells, allowing their destruction by different means, such as infrared sources, biochemical agents, etc.
• Nanobio innovations strongly promote the integration of different applications into advanced technology platforms for life sciences research.
Nanobiosensing/Cellular Imaging
• Integrated nanobiosensors indicating the presence of particular molecules or biological structures in microdevices represent important improved capabilities for life sciences and healthcare.
• Cellular imaging represents one of the most promising tools for early diagnostics and personalized medicine. Enhanced imaging technologies by using quantum dots and nanobiomarkers accelerate the pace of future medicine.
Among the most lucrative markets for nanobiotechnologies, medical and life science applications appear first. The advent of nanobiotechniques by hand of LOC devices and nanoenabler drug delivery methods, along with a number of novel experimental medicine and clinical/preclinical studies for early clinical trials, have become micro and nanofluidic techniques as the starring technologies for next-decades developments.
The underlying key of such prosperous development regarding nanobiotech applications comes from the reality that nanoscale particles, including nanoscale devices, are 100 to 10,000 times smaller than human cells. Indeed, nanoparticles are similar in size to large biological molecules, including as enzymes and bioreceptors.
According to this, three principal areas of interest have been mostly related to the promising development of nanofluidics and nanobio devices:
• Disease diagnosis and screening
• Drug delivery systems
• Health monitoring
CORE GROWTH OPPORTUNITIES
Biopharmaceuticals/Next-Generation Biomarkers
• Integrating new technologies into industry value chain at several levels performing ultrahigh-throughput and parallel analysis at the single-molecule level.
• Accelerating the pace of drug discovery using high-throughput techniques.
• Enhancing target selection, lead identification, preclinical tests, clinical trials, chemical synthesis, formulations studies, and product management.
Oncology/Nanoarrays Developments
• Evaluating large number of chemical structures against hundreds of biological targets through novel miniaturization and massively parallel experimentation methods.
• Integrating different functional units for sampling, sample pretreatment, sample transport, biochemical reactions, analyte separation, product isolation, and analysis in a continuous flow manner.
High-Sensitivity & High-Precision Instrumentation
• Developing nano/microfluidic devices with high analytical throughput rates.
• Detecting DNA translocation events through solid-state nanoarray devices as a proof-of-concept that illustrates ultrahigh sensitivity and specificity.
• Interfacing micro/nano and macro-technologies through novel, ease-handling, robust, multiplex, and cost-effective nanobiochips.
MARKET OVERVIEW & PROSPECTIVE LANDSCAPE
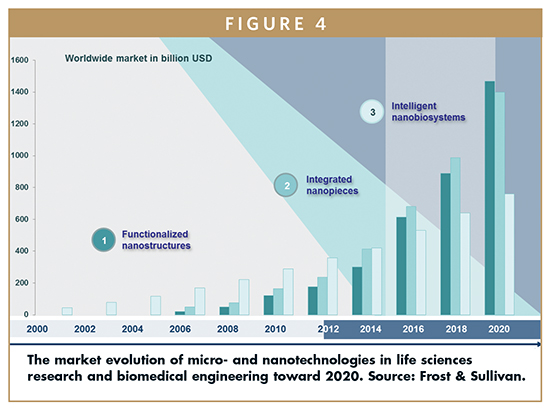
Addressing the substantial impact of nanobiotech developments, and especially nanofluidics and bioNEMS on the world’s economy, a prospective landscape of market volumes and dynamics constitutes a crucial indicator. Although, the number of industries and the synergistic approaches among a wide range of disciplines, along with the complexity of players in the market, such a quantitative approach represents a matter of great concern.
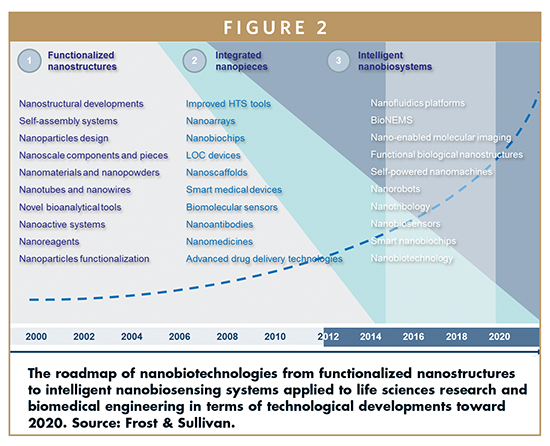
By screening the investigations and industry breakthroughs evidenced throughout the past decade, the numbers of inventions and innovative ideas, as well as the number of funding programs and human resources involved in the broad spectrum of nanotech-based markets, have all increased by an average annual rate of more than 27%. Indeed, following with the aforementioned defined three levels, that is:
• Functional nanostructures
• Integrated nanopieces
• Smart nanobiosystems
Applied to the life sciences and biotech industry, the total worldwide market reached about $800 billion in 2011, expecting over $1.8 trillion by 2015 and $3.6 trillion by 2020. Interesting, the two last categories have begun to grow as marginal technologies. Nevertheless, their increment throughout the past 5 years, supported by the previous growth of the first category, has resulted in a real breakthrough.
INVESTMENT
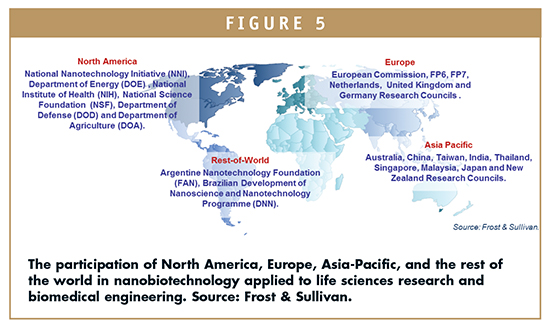
Global public and private investments in research and development (R&D) have grown by 40% throughout the past years. Worldwide expenditures have been disaggregated into venture capitals, government grants, regional programs, and private investors. Important differences are being evidenced by regional landscape.
Remarkably, North America possesses a strong participation of private sectors, along with government grants, whereas Europe holds a strong position over regional programs. Asia-Pacific region shows the strong involvement of government in R&D decisions, the same as most other regions, such as South America.
Due to fluids exhibiting unique physical behaviors at nanometer scales, different from those present in larger structures, nanofluidic structures have successfully found a broad spectrum of applications. Such variety of technologies range from analytical separations, protein or molecular building, and RNA/DNA manipulation, among many others.
Naturally, nanofluidics developments not only involve an increasing number of applications emerging but also promote the appearance of a wide variety of innovative fabrication methods and technologies enabling the successful development of these applications. Bionano-electromechanical systems (BioNEMS) applications, on the other hand, experience a similar behavior, being even more significant the role of the enabling technologies.
Strong discipline focus is put on life sciences and bioengineering disciplines, expecting a central role of nanofluidics and bioNEMS in future medicine. The complexity of the technology and the access to appropriate equipment, as well as the development of tech enabler technologies, make the advancements of nanofluidics and bioNEMS limited in comparison with microfluidics technologies, for instance.
First-level equipment regarding nanomanufacturing, clean rooms, pumping techniques, and energy need to be set up, which constitutes a limitation for R&D activities. On the other hand, the successful integration of individual components and surfaces for nanofluidics devices and bioNEMS still challenges researchers and scientists.
Despite these limitations, strong evidence set nanofluidics and bioNEMS developments as one of the most promising areas of research worldwide. The relevance of nanobiotech developments is clear when analyzing both governmental and private investment and funding trends in nanosciences related to these particular technologies.
A solid intellectual property (IP) position provides R&D institutions and collaborative actors with a stronger strategy to face business development activities and design new methodologies to transfer the technology to industry sectors through an optimal resolution.
TECHNOLOGY CROSS-POLLINATION
As a physics phenomenon, nanofluidics has been defined several decades ago. However, as a discipline, nanofluidics has emerged only in the past few years with the advent of micro and nanotechnologies.
Nanofluidic channels range from a random network of pores to well-defined and oriented networks. In this regard, advance manufacturing techniques allowing building nanostructures with the desired properties, practically selected according to the geometry of a device and its application, have begun to play a highly dynamic role.
Beyond the structure, nanofluidic systems and devices constitute powerful tools, applicable not only to study fundamental nanoscale science, but also for practical biochemical and clinical applications.
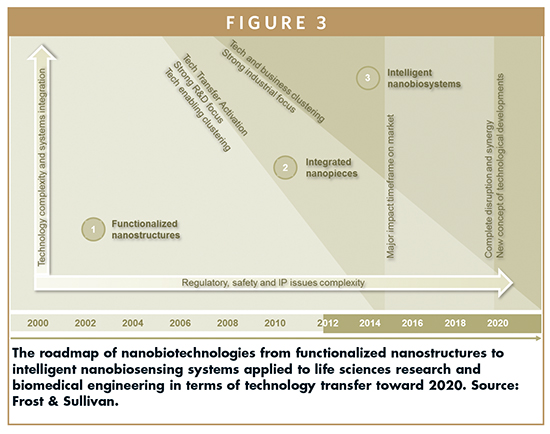
The convergence of a plethora of technologies suggests the creation of new technology clusters leveraging the synergy among several disciplines. Predictions for 2020 claims for a total restructuration of both tech and business clusters and niches favoring innovation and technology translation from R&D institutions to industry sectors. This convergence can be strongly evidenced in terms of products.
A direct correlation with business development activities is exhibited by evaluating the evolution and dynamics of business models in the time, taking as a reference research, development, and tech transfer tasks.
FINAL REMARKS
The innovation landscape in nanofluidic devices and nanobiosensors is unlimited. Stakeholders involved in innovation processes, technology transfer, and market incorporation are numerous. Stringent regulations and costly device safety procedures delay the efforts to commercialize MEMS-based medical devices. The timeline from product development to commercialization is long for medical devices than most consumer products. To bring a bioMEMS device to the market, manufacturers need to adhere to various regulations in terms of the manufacturing, safety, and sustainability of the device.
Among the disease diagnosis and screening applications, LOC devices offer a broad spectrum of diagnostic functions, allowing achievable similar results from a complete medical or clinical laboratory, is of the most notable. Similarly, nanobiosensors based on nanotubes, wires, magnetic particles, and semiconductor crystals, including quantum dots, come with remarkable advantages for the near future of medicine and life sciences. Analogously, novel medical imaging techniques can be significantly benefited from the advent of nanobiotechnologies.
Drug delivery systems, also coined as nanoenabler drug delivery systems, include a great variety of nanocapsules, dendrimers, and fullerene-based structures aiming to address a controlled- and sustained drug-release mechanism. Remarkably, the past decade has evidenced a highly focused attention of both portable medical devices for diagnostics and drug delivery systems for therapeutics, not only in developed countries, but also for countries lacking from appropriate drug storage capabilities and distribution networks, such as diverse developing countries.
Health monitoring, on the other hand, could experience a significant change from the appearance of nanobiodevices. The periodic evaluation of physiological variables in a cost- and time-effective manner, also facilitating comfort and building capabilities, through novel POC devices that significantly help to address current issues around healthcare burden like aging population and personalized medicine, among many others.
To view this issue and all back issues online, please visit www.drug-dev.com.

Cecilia Van Cauwenberghe is a Senior Industry Analyst with Frost & Sullivan’s global Technical Insights practice. She has over a decade of professional expertise in chemical and biomedical engineering arenas, which include R&D activities in several well-renowned universities and multinational companies. As part of her experience, Cecilia has been involved in a variety of multidisciplinary projects embracing different emerging technologies and establishing strong long-standing working relationships. Cecilia’s knowledge base encompasses biopharmaceutical and biochemical modeling, simulation and optimization; microelectronic implantable devices design; and research in metabolic syndrome and cardiovascular diseases applications. Cecilia holds master and doctoral studies in Chemical and Biomedical Engineering.
Total Page Views: 3690