Issue:March/April 2026
COMBINATION PRODUCTS - Early Decisions to De-Risk the Transition to Combination Products
INTRODUCTION
The combination product market has seen a significant amount of growth, driven primarily by the rise in chronic disease indications, the demands for self-administered therapies, and technological advancements. The use of combination products simplifies the process of drug administration for the patient and/or the caregiver and, in some cases, allows the patient to receive treatment at home rather than having to travel to a medical facility. However, there are many challenges that need to be navigated as one considers the transition from a vial system to a needle-based combination product.
The US FDA defines a combination product as comprising two or more constituents and classifies them into the following four types:
- Drug + Device
- Biologic + Device
- Drug + Biologic
- Drug + Biologic + Device
Each constituent retains its regulatory status. For example, a combination product comprised of a drug or biologic, and a device must meet the regulatory requirements of both the drug or biologic and the device, with the filing center being based on the primary mode of action of the combination product, either CDER/CBER or CDRH.
In the EU, a combination product can also be a medicinal substance with an integral or non-integral (co-packaged or referenced) medical device and the regulatory filing must include both the medicinal substance filing and the device filing.
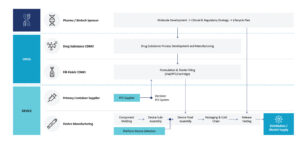
COMBINATION PRODUCT DEVELOPMENT CHALLENGES
An important part of combination product development is compiling the regulatory submission. Without approval from regulatory bodies, products do not make it into regulated markets. The regulatory landscape is ever-changing within the combination product development space and regulators now have a lot of direct combination product subject matter expertise. As a result, there is an expectation from regulators that pharma companies that file a combination product application have a full understanding of the drug/ biologic constituent as well as an equally full understanding of the device constituent, which is not always their forte.

The second challenge is that formulations and therapies are increasingly complex and drug stability can be highly dependent on its interaction – or hopefully lack of interaction – with the primary container and with its flow path. As such, the compatibility and performance of the related system critical components used with the drug/ biologic constituent must be understood. Unlike vial systems, combination products may have moving parts and often require lubrication or other modifications that can affect drug stability.
Thirdly, there is the challenge of delivering great patient experience. The growing preference for self- or home-administration means that more medications are integrated with devices. The challenge is that devices must be easy for the patient to use and empower them to receive medication at their convenience, while simultaneously addressing costs associated with therapy administration. However, devices must be easy to use and empower patients to receive medication at their convenience while simultaneously addressing costs associated with therapy administration.
Finally, with more competition among drugs treating the same disease, as well as the growth of generics and biosimilars, there is a greater need for product differentiation, which can be achieved through an improved drug delivery system. A focused approach to moving from a vial format to a device driven combination product is recommended with three critical de-risk factors which include: the Plan, the Product, and the Patient Experience.
DE-RISKING THE PLAN
Key Considerations
- Effective lifecycle management
- Shelf-life study history (accelerated/real-time)
- Supplier regulatory submission support
- Simplified data package availability
- Subject matter expert support
- Direct risk assessment linkages
- Combination product testing support
Fundamentally, drug developers want to avoid introducing risk as they journey from clinical-milestone to clinical-milestone. There is a lot of uncertainty around combination products, and companies’ common reaction to this uncertainty is to go to market with a simple, known, off-the-shelf delivery format not tailored to the molecule or to patient needs. However, simply avoiding complex product development is not patient-solution driven and will result in missed opportunities. A better approach is to begin with the end in mind and focus on the plan beginning with Lifecycle Management.
Starting with a simple delivery format and delaying Lifecycle Management until after commercial success is risky because companies often realize too late that their drug is at a competitive disadvantage and begin Lifecycle Management efforts only when it is too late. The results are higher overall costs, unfulfilled market potential, and, possibly, losing the momentum of being the first on the market. Biosimilar and Generic companies might underestimate the relentlessness of the originators to protect their brands. Thus, they are surprised to see that by the time they get their approval to launch the biosimilar or generic, the originator already converted the market to an advanced presentation and the biosimilar or generic is left chasing-the-innovator.
Partnering with suppliers that have component and system shelf-life testing history and knowledge, and with a proven track record in successful regulatory submissions, will help minimize surprises. When evaluating partners, make sure you understand the partner’s credentials in the field. Is your drug going to be the partner’s first to make the entire journey to regulatory submission and to commercialization or did the partner do this already for another drug. Does the partner have meaningful
platform data that could be leveraged into your development to save time and resources? If the supplier already has component extractables and device performance data packages, you can hit the ground running with application-specific testing, like leachables and drug delivery.
It is critical to partner with industry subject matter experts to establish risk-assessment-linkages from components to the combination product. When these linkages are established, it is easier to avoid surprises, shorten timelines, improve operational efficiencies that reduce costs, and increase the probability of regulatory success. Finally, you want a device partner who can also provide performance testing support for the final drug-device combination.
All of this means that the combination product can get to the patient faster with a competitive advantage, serve diverse market needs and most importantly, deliver a better patient experience, which also, naturally, leads to improved adherence to the drug regimen.
DE-RISKING THE PRODUCT
Key Considerations
- Patient and drug requirements
- Component knowledge
- Material, chemical, physical interactions
- Proper component selection
- System integration
- Suitability assessments
- Risk management
- Robust supply chain
- Experienced partner(s)
De-risking the product requires a thorough understanding of user requirements to know exactly what the product needs to do. By leveraging in-depth component knowledge from manufacturers – particularly regarding materials’ chemical and physical interactions – system integrators can navigate proper-component selection to develop an optimal device design. These resources within the supply chain must possess the knowledge and expertise to assess suitability from the component level all the way through to the whole device constituent part of the combination product. They must also have established risk management expertise to connect component level assessments to higher level risk assessments of the complete combination product.
DE-RISKING THE PATIENT EXPERIENCE
Key Considerations
- Self-administration
- Ease-of-use
- IFU simplicity
- Minimize potential misuse
- Dose delivery optimization
- Pain threshold
- Adherence to therapy
- Direct component impact
A proactive approach to obtaining first-pass regulatory approvals must de-risk the patient. For example, as therapies become more complex and shift towards home administration, pharma companies are under pressure to bring more intricate devices to market that are also easy for patients to use.
But this isn’t easy to do with constantly changing regulations and expectations that place more responsibility on pharma companies to prove that not only is their drug constituent safe and effective (which is their expertise), but to also show that the device constituent is safe, effective, reliable and with adequately designed-in human factors consideration, which are not aspects that are typical pharma company areas of expertise.
Such systems intended for home use need to require minimal thought from the user. If instructions are unclear or confusing, or if the system has not been thoroughly tested for real-world administration – including potential misuse – this can result in a poor patient experience, patient pain, adherence challenges, compromise the therapy and reduced overall market acceptance – possibly even leading to product recall. Some of these factors can be a direct result of the component selection discussed earlier.
SUMMARY
In summary, when transitioning from a vial to a combination product system, the best approach is to have a holistic de-risk strategy with an intentional focus on the Plan, the Product and the Patient Experience. It is vital to select partners with in-depth understanding of the evolving regulatory landscape, that have a systems mindset, that have industry partnerships and that have subject matter expertise in areas required to establish drug-device suitability. With this approach, it is easier to avoid surprises, shorten timelines, improve operational efficiencies that reduce costs, increase the probability of regulatory success, and ultimately reduce risk to the patient. This approach will get the combination product launched and into the market faster while delivering a safe, effective, and positive patient experience.
When it comes to transitioning pharmaceutical products into a combination product system, West Vantage™ offers contract manufacturing solutions that enable customers to select the right combination product and facilitate design, system integration, analytical testing, manufacturing, filling and assembly. Additionally, West Vantage™ offers services to assist in regula tory dossier compilation and filing, as well as analytical testing via laboratories in Exton, PA and Waterford, Ireland to support the design verification and release testing of combi nation products. Supported by West’s century-long leadership in containment systems, West Vantage™ offers the capacity, technical discipline, and operational excellence required to as semble and prepare complex delivery sys tems for clinical and commercial distribution. To learn more, visit WestPharma.com/Services.
Vantage is a registered trademark of West Pharmaceutical Services, Inc. in the United States and other jurisdictions.

Dr. Mike Ulman has over 17 years of experience in the pharmaceutical industry. For the past year, he has worked in Regulatory Affairs, focusing on how chemical and environmental regulations affect the pharmaceutical packaging business, while for the previous three years, he spearheaded the development of performance testing methodologies for combination products as the Technology Manager for Packaging and Delivery Systems, both at West. Prior to joining West, he spent 13 years at Piramal Critical Care, holding various technical roles in inhalation anesthetics, and nine years as a chemist at Air Products. He holds a PhD in Chemistry.
Total Page Views: 107