Issue:March/April 2026
THERAPEUTIC FOCUS - Advancing Adjunctive Therapies for Depression & OCD Using Translational Pharmaceutics®
ABSTRACT
Depression affects an estimated 5% of adults globally, and in the US, depression diagnoses have risen 33% since 2013; twelve-month depression prevalence is 10.7%; and healthcare spending by a patient suffering from depression is twice that of the average patient.1 Unfortunately, less than one-third of patients respond adequately to first-line serotonin reuptake inhibitor antidepressants (eg, Prozac®) therapy.2 Current next-line options are limited by modest efficacy and safety concerns.3 For patients who do not respond adequately to first-line antidepressants, new next-line options are urgently needed.
Quotient Sciences (Nottingham) partnered with Evecxia Therapeutics to support the development of EVX-101, an investigational adjunctive drug for depression and obsessive-compulsive disorder (OCD) responding inadequately to first-line antidepressants. EVX-101 is a first-in-class drug acting via Serotonin Synthesis Amplification. Using Quotient Sciences’ Translational Pharmaceutics® platform, the team developed and manufactured the drug product and conducted an adaptive clinical study to optimize performance against the target product profile.
A gastro-retentive (GR), bilayer modified-release (MR) EVX-101 tablet formulation combining 5-hydroxytryptophan (5-HTP) — the natural immediate serotonin precursor — with low-dose carbidopa (enhances 5-HTP’s bioavailability) was developed and manufactured for clinical testing. This formulation approach effectively addressed the inherent challenges of 5-HTP’s short half-life, narrow absorption window, and low bioavailability.
The two-part clinical study involved a sipping protocol to confirm the GR strategy and to define the carbidopa dose range (Part 1) and a formulation optimization study using a 2-dimensional design space to vary the release rate of the two drugs and the GR retention time (Part 2). The formulations were radiolabeled with not more than 1 MBq 111indium allowing gamma scintigraphy to be used to assess in vivo formulation performance. The optimized formulation was then progressed to a Phase 1 program, which included single ascending dose (SAD) and multiple dose titration (MAD) study in healthy volunteers treated with escitalopram.4
The program achieved significant time savings compared to conventional formulation optimization strategies with two Phase 1 trials completed in a timely manner. The following reviews the Phase 1 study design and clinical outcomes, leveraging data presented at the 2023 AAPS Annual Meeting.5,6
INTRODUCTION
Inadequate response to antidepressant treatment is a significant issue for many patients with depression.2,3 Enhancing brain extracellular serotonin (5-HT) levels beyond those produced by serotonin reuptake inhibitors may treat depression more effectively.7 Adjunctive treatment with 5-hydroxytryptophan (5-HTP), the natural precursor to 5-HT, can amplify brain serotonin.8 However, 5-HTP absorption is restricted to the upper intestine, bioavailability is low, and 5-HTP has a short half-life (t½), making 5-HTP a poor drug.
To overcome these pharmacokinetic (PK) limitations, a gastro-retentive (GR) modified release (MR) formulation was required to maximize the absorption window for sustained delivery exposure. Carbidopa inhibits amino acid decarboxylase, the enzyme that converts 5-HTP to 5-HT in the intestine, resulting in decreased first-pass metabolism and increased 5-HTP bioavailability. The effective dose of carbidopa was unknown. The goals of each part of the study were as follows:
Part 1: administering 5-HTP as a sipping protocol to simulate GR formulation input and to demonstrate proof of concept (PoC) for the GR formulation. Additionally, the flexibility to adjust carbidopa dose to define the dose range for the GR formulation was considered.
Part 2: developing and optimizing EVX-101 as a novel GR MR 5-HTP/carbidopa bilayer tablet formulation and conducting PK and safety assessments. The target product profile (TPP) was to achieve a twice a day (BiD) dosing regimen with a steady state Cavg (ssCavg) of >100 ng/mL.
Quotient Sciences’ Translational Pharmaceutics platform was used to conduct the study, integrating drug product formulation development and GMP manufacturing with clinical dosing activities.

METHODS
The conventional paradigm for the development of GR dosage form is to screen a range of prototypes in preclinical species, usually dogs, and use this data to select two or three fixed compositions to manufacture and subsequently dose in a human PK
study. The inherent risk of this model is the acknowledged lack of predictability of animal bioavailability to that which might be expected in humans. For GR products, this is particularly true given the significant physiological and anatomical differences between species.
When Translational Pharmaceutics is applied in drug product optimization, multiple formulation technologies or prototypes can be assessed during a clinical study. Formulations are then optimized based on clinical data, addressing the issue of poor preclinical predictability. Small batches are manufactured immediately prior to dosing, saving time in formulation scale up and stability data generation. Interim data reviews after each dosing period assess safety, tolerability and PK to determine the next technology or prototype to be manufactured and evaluated, typically on a 2-3 week cycle time.9
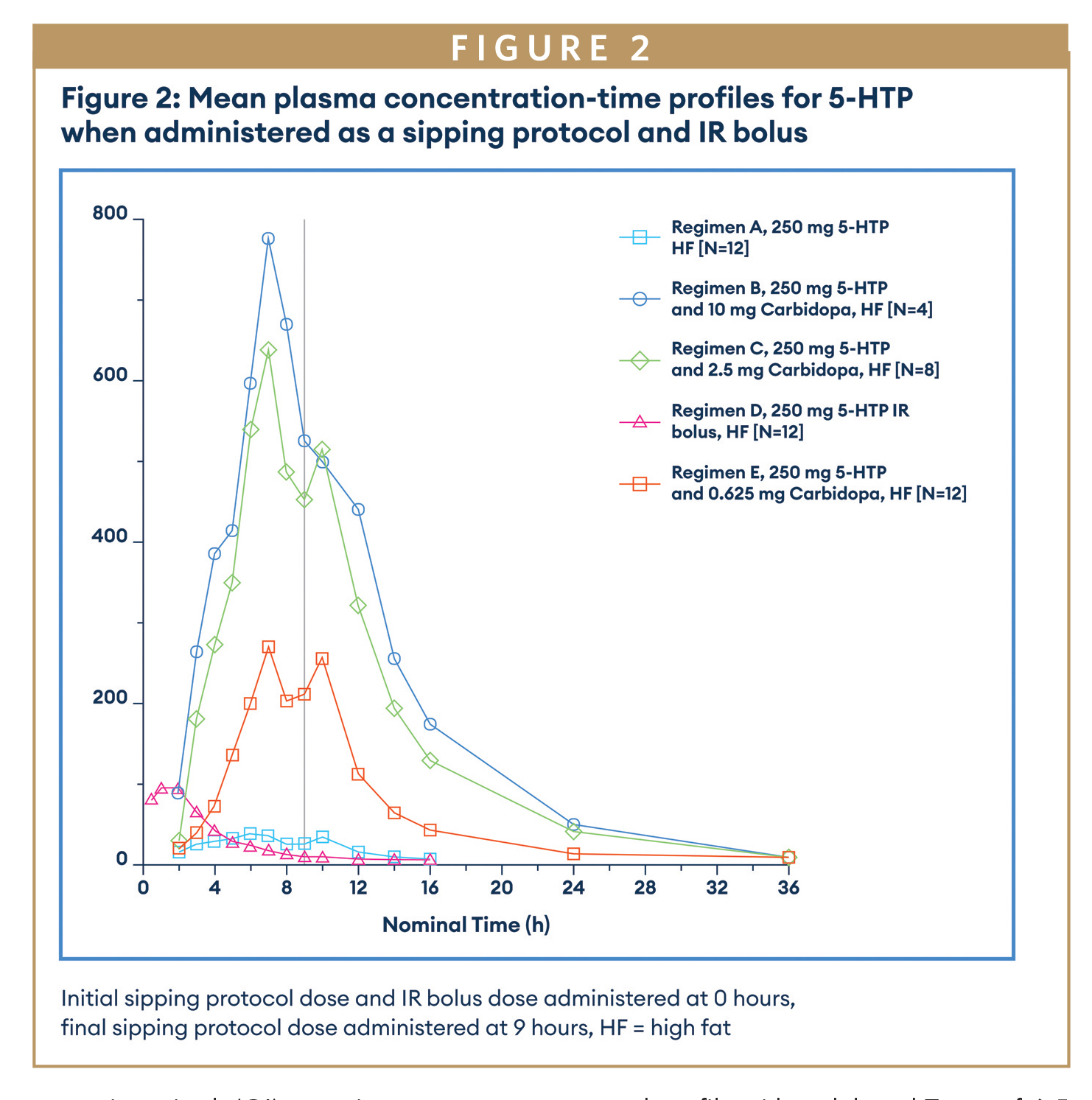
In Part 1, 12 healthy subjects were dosed in a 5 period sequential study, with each subject receiving a fixed dose (250 mg) of 5-HTP as an immediate release (IR) bolus dose, a sipping regimen (divided into 10 aliquots administered over a 9-hour period) and 5-HTP with three different dose levels (0.625 mg, 2.5 mg, and 10 mg) of carbidopa as sipping regimens. Both drug products were administered as solution formulations.
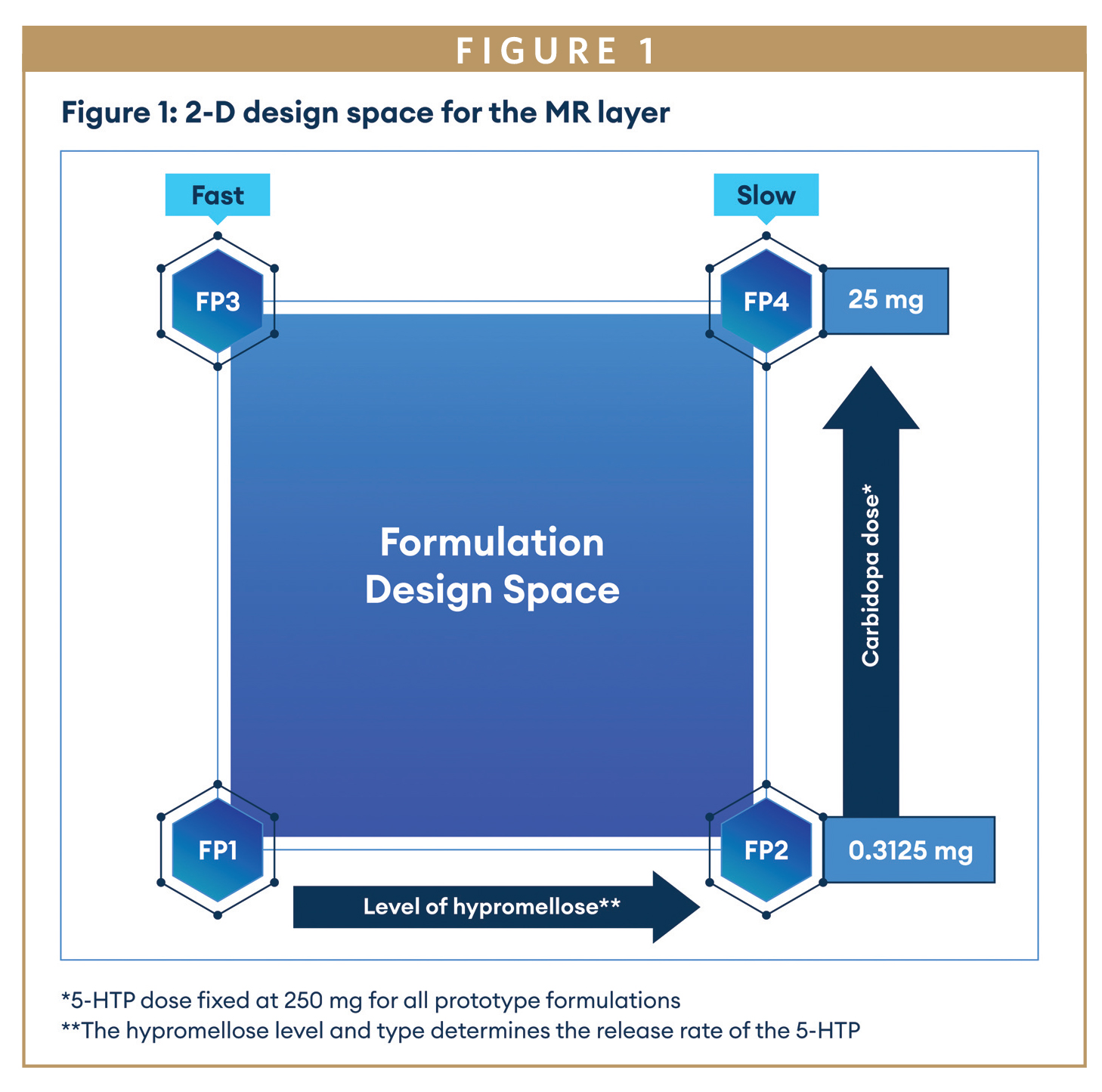
In Part 2, a new cohort of 16 healthy subjects received five sequential dosing periods. Quotient Sciences developed a bilayer GR/MR tablet containing a fixed 5-HTP dose (250 mg) and a two-dimensional design space (Figure 1) to enable variations in carbidopa dose (0.3125 mg – 25 mg) and the level of hypromellose (K4M, 7%-18%) and K100M (0%-25%) to control release rate of 5-HTP and carbidopa (target 80% release 8.5 to 12 hours. The formulations were radiolabeled with 111indium, allowing gamma scintigraphy to be used to assess in vivo performance based on gastric emptying (GE) time, gastrointestinal (GI) transit parameters, and initial and complete tablet disintegration (ITD and CTD). The carbidopa dose-response relationship to 5-HTP exposure was also assessed. In periods 1-4 subjects received GR prototype formulations in the fed state following a high fat/high calorie breakfast, with period 5 dosed in the fed state following a moderate fat/moderate calorie breakfast.
For both study parts, interim decisions occurred between dosing periods, whereby PK, safety, and scintigraphy data (Part 2 only) were reviewed to assess formulation performance and decide what formulation prototype to dose in the subsequent period.
RESULTS
When 5-HTP was delivered as a sipping protocol in Part 1, it gave the expected profile with a delayed Tmax of 6.5 hours compared to the bolus IR dose Tmax of 1 hour and also reduced exposure. Carbidopa had the anticipated effect on 5-HTP exposure (Figure 2) increasing exposure and t½, with a 2.5 mg dose resulting in a 16-fold increase in 5-HTP exposure. 10 mg carbidopa resulted in additional increases in 5-HTP exposure; however, there was a limited data set for the 5-HTP 250 mg and carbidopa 10 mg regimen due to subjects withdrawing due to adverse events (AEs), and hence the data should be viewed with caution. Carbidopa plasma levels were only above the LLOQ (1 ng/mL) for the 10 mg carbidopa dose. Note, all the 5-HTP and carbidopa sipping regimens dosed, achieved or exceeded the steady state target plasma concentrations for 5-HTP. However, when switching to a GR formulation, the 5-HTP exposure is predicted to be lower than when compared with the sipping regimen data, and hence a wider dose range for carbidopa was anticipated to be required.
The data from Part 1 allowed the dose range for carbidopa to be set at 0.3125 mg to 25 mg for the GR prototype formulation to be dosed in Part 2 of the study. The PK profile achieved by dosing the sipping protocol with 5-HTP and carbidopa confirmed PoC for a GR formulation.
During the GR bilayer formulation development, it was observed that the order of the two layers being compressed can also affect MR layer’s surface area and the drug release rate. Hence, it is a critical factor to achieve the desired release rate. When the bilayer tablets were pressed in the order of GR-to-MR, the drug release rate was faster and more sensitive to the polymers used in the MR layer. The bulk density of the GR layer was less dense compared to the MR layer. Therefore, the decision to compress the MR layer first was deemed suitable to improve the manufacturability of the formulation. This finding is useful for development of bilayer MR tablet to achieve the required drug release profiles and is also critical for downstream scale up process development as the sequence of compression will impact on the release profile.
In Part 2, a new cohort of 16 healthy subjects each received GR prototype 1 (0.625 mg carbidopa and 250 mg 5-HTP, target release rate 80 % at 8.5 hours) in the fed state in period 1. GR prototype 1 produced a similar 5-HTP exposure to the 250 mg 5-HTP IR bolus dose in Part 1 of the study, suggesting a higher dose of carbidopa is required to achieve the target 5-HTP exposure. Imaging of the radiolabeled formulation within the gastrointestinal tract showed that GR formulation had a mean gastric emptying time of 5.863 hours, initial tablet disintegration of 0.691 hours, and complete tablet disintegration at 18.501 hours. The in vivo imaging showed that adequate gastric retention was achieved from the GR formulation with the target 80 % release at 8.5 hours (Figure 3). Therefore, the release rate properties for 5-HTP (target 80 % release at 8,5 hours) were fixed for future GR prototypes and exposure modified by changing the carbidopa dose level. GR prototypes with 2.5 mg, 5 mg, and 15 mg carbidopa were also dosed to explore the effect of increasing dose levels on 5-HTP exposure (Figure 4). Exposure with all these dose levels increased 5-HTP exposure, and it was predicted (using modelling data) that these prototypes would meet or exceed the target ssCavg on multiple dosing. Carbidopa plasma levels were only above the LLOQ (1 ng/mL) for the 15 mg carbidopa dose. The GR properties (GE time) for all 4 GR prototype formulations dosed were consistent as assessed by scintigraphic imaging, and when a prototype was dosed with a moderate fat meal, gastric emptying time was reduced compared to when dosed with a high fat meal, as expected with the reduced calorie and fat content, however the gastric emptying properties were still acceptable for a BiD GR formulation dosing regimen.
SUMMARY
Translational Pharmaceutics allowed emerging clinical data to be used within a clinical study to validate PoC for a GR formulation, refine the carbidopa dose range, confirm MR release rate and GR properties, and identify an EVX-101 GR
tablet formulation to achieve the target profile of BiD dosing and ssCavg >100 ng/mL. The optimized EVX-101 GR tablet formulation was safe and tolerated within the target ssCavg range, and was recently used to successfully complete a single and multiple ascending dose study.4
REFERENCES
- Hasin DS, Sarvet AL, Meyers JL et al. Epidemiology of Adult DSM-5 Major Depressive Disorder and Its Specifiers in the United States. JAMA psychiatry. 2018; 75(4): 336-346.
- Trivedi et al 2006 [https://pubmed.ncbi.nlm.nih.gov/16390886/].
- Berman et al 2007 [https://pubmed.ncbi.nlm.nih.gov/17592907/].
- Carpenter, D. et al, Poster T30. 2023 Annual Meeting of the American Society of Clinical Psychopharmacology.
- 2023 AAPS Annual Meeting clinical poster.
- v2023 AAPS Annual Meeting – hydrogel poster.
- Jacobsen et al 2016 [https://pubmed.ncbi.nlm.nih.gov/27692695/].
- Turner et al 2006 [https://pubmed.ncbi.nlm.nih.gov/16023217/].
- DiMasi, J.A., Wilkinson, M. Ther Innov Regul Sci 54, 1453–1460 (2020).

Dr. Jacob Jacobsen serves as CEO of Evecxia Therapeutics, a company he co-founded and spun out of at Duke University in 2019. Evecxia is dedicated to realizing the therapeutic potential of Serotonin Synthesis Amplification, a novel pharmacology for the treatment of psychiatric disorders. Prior to joining Evecxia, he was a scientist at Duke University and Duke-National University Singapore, where he was the driver of a cross-disciplinary team executing seminal research on Serotonin Synthesis Amplification. He also executed several basic and applied neuroscience projects that resulted in multiple first and co-author publications in journals of repute. Prior to Duke, he spent 8 years in pharma-biotech in Copenhagen, Denmark, ie, NeuroSearch and Lundbeck, where he executed drug discovery and target validation projects in preclinical models. He is well-published, including in the highest-ranking journals in the field of Psychiatry, and is an inventor on all Evecxia issued and pending patents. He earned his PhD in Neuropharmacology and MS in human biology from the University of Copenhagen.


Dr. Bret Berner has 47 years of industrial experience in drug delivery. After earning his PhD in Neurosciences/Physical Chemistry at UCLA, he joined Procter & Gamble Co. studying surfactants, diffusion, and skin permeation. He was Director, Basic Pharmaceutics Research at Ciba-Geigy, followed by Vice President, Development, at Cygnus Therapeutics, and then Chief Scientific Officer at Depomed. For over 15 years, he has been a pharmaceutical consultant in drug delivery, formulation, and pharmacokinetics. He holds over 70 US patents, authored more than 80 publications, and edited 3 books on drug delivery.
Dr. Vanessa Zann has over twenty-five years industry experience providing expert biopharmaceutic support to drug discovery, early drug development and clinical program design. Vanessa holds a PhD in Pharmaceutical Science from Aston University, and completed postdoctoral research in buccal transport, before joining AstraZeneca as a permeability expert in the Pharmaceutical Development department, leading the global Caco-2 facility and selection of new chemical entities with appropriate biopharmaceutical properties. Since joining Quotient Sciences in 2012, Vanessa has provided study design and scientific leadership to over 100 clinical pharmacology and drug product optimization studies across a wide range of applications including first-in-human, drug-drug interaction and development and optimization of solubility enhanced and modified release formulations. She led the implementation of modelling and simulation services and application of in-vitro characterization strategies to Quotients development programs.
Total Page Views: 122















