Issue:November/December 2017
NEXT-GENERATION SEQUENCING - Emerging Clinical Applications & Projections to 2022
INTRODUCTION
The growth curve for clinical applications of next-generation sequencing (NGS) is approaching an upward trajectory as existing applications gain traction and new ones begin to establish a foothold. BCC Research found that principal drivers of market growth in the clinical NGS industry include precision medicine and genomics initiatives; penetration into large market economies such as China; and the emergence of high-potential applications in average-risk noninvasive prenatal testing, preventive genetic screening, and early cancer detection.
Several unique aspects of NGS make it a desirable technology for clinical applications. Because NGS platforms can sequence entire genomic regions or even whole genomes, a single test can examine hundreds or thousands of clinically important genetic variations. An NGS platform also allows companies to expand their menu of disorders and diseases over time, after initial launch of a test. This strategy has been used in reproductive health applications, for example, launching a test to initially screen for aneuploidies and later expanding it to include screening for additional genetic variants.
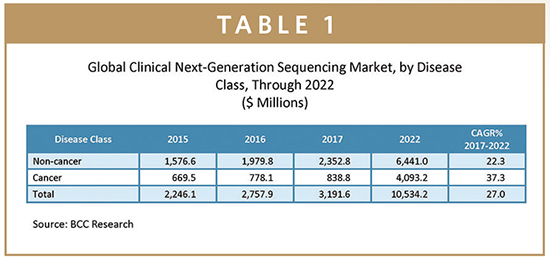
The global clinical NGS market is estimated at approximately $3.2 billion in 2017 and is forecast to increase at a compound annual growth rate (CAGR) of 27% to reach $10.5 billion in 2022. Non-cancer applications accounted for an estimated $2.4 billion in 2017 and are projected to grow at a CAGR of 22.3% to reach $6.4 billion in 2022. Cancer applications, valued at roughly $839 million in 2017, have an anticipated CAGR of 37.3%, putting this segment at close to $4.1 billion in 2022. Important cancer applications include screening and early detection, diagnosis, therapy guidance, and monitoring.
NGS is often more accurate and reliable than existing diagnostics. The following sections examine the main applications for NGS diagnostics that are commercially available or forecast to emerge in the next 5 years.
CANCER APPLICATIONS
The main applications of NGS diagnostics in cancer are tumor sequencing, familial screening, and monitoring for cancer recurrence. It is becoming increasingly apparent that cancer is a genetic disease. Knowing the genetic profile of a tumor can change the way that cancers are treated, eg, by determining which drug is given to a patient. There is a move toward classifying and treating cancers on the basis of their genetic makeup rather than their location in the body. For example, trastuzumab is effective for treating breast cancers in which the gene ERBB2 is present. However, some colon cancers also overexpress this gene and may also be treatable with trastuzumab. Cancer diagnostics that can genetically profile a tumor could thus be clinically valuable and influence a physician’s treatment decision. This is a critical driving force behind NGS-based tumor sequencing diagnostics. The key issue for this application is establishing a clinical connection between the mutation profile of a given tumor and an effective therapy. It is believed, however, that many large-scale sequencing projects are addressing this issue and significant progress is being made.
Current applications of NGS in cancer are focused on discrete sets of genes. However, BCC Research believes that whole-exome and whole-genome sequencing will increasingly gain traction as sequencing costs fall and the value of testing for only a single gene (or several genes) declines. Longer term, it is expected that most tumors will be completely sequenced so that physicians can have a complete set of genetic information when treating patients.
The second application of NGS in cancer is familial screening offered to relatives of patients with cancer or individuals otherwise at risk. Relatives of patients with cancer could be at high risk of developing cancer, and genetic testing among a broader population such as this could reduce the mortality rate and overall costs of the disease. The key driving force for this application is a reduction in sequencing costs to the point where there are significant benefits versus costs for NGS tests.
Another driving force for familial screening is breakout into the general population. There is evidence that many people who have no history of cancer in their family can still carry cancer-risk mutations. It is unlikely that the general population of at-risk people will undergo NGS-based screening tests without insurance reimbursement. Therefore, both sides of the cost-to-benefit-ratio must be improved (ie, NGS costs must continue to decrease and clinical validity must be established).
The third application of NGS for cancer through 2022 is monitoring for cancer recurrence. This involves the capture and sequencing of low levels of circulating tumor cells in peripheral blood. There have recently been outstanding technical developments in this area, and continuing progress in this field will drive this application. BCC Research estimates that by combining state-of-the-art circulating tumor cell capture methods with whole-tumor NGS, cancer monitoring applications will reach commercial viability within the next 5 years.
MENDELIAN DISORDERS APPLICATIONS
Mendelian disorders (rare genetic disorders) are difficult to diagnose. Diagnostic odysseys occur when an individual has a rare genetic disorder and undergoes multiple expensive, invasive clinical procedures to determine the cause. These cases occur frequently in clinics that evaluate children for things such as cognitive impairment, neuromuscular disorders, or congenital anomalies. Diagnostic odysseys often include serial molecular testing of one or a few genes, running up the costs of diagnosis.
Many of these rare genetic diseases are due to a single-gene mutation in the genome. They are referred to as Mendelian disorders because they comport with the inheritance pattern first discovered by Gregor Mendel. BCC Research believes that there are as many as 25 million individuals in the US who have inherited Mendelian disorders. Some of these are well understood, including cystic fibrosis and muscular dystrophy, but others are much rarer. There are approximately 6,000 of these very rare disorders, and only half of them have been identified.
NGS is changing the diagnostic paradigm in these cases because it can produce a correct diagnosis faster and more cost effectively than conventional approaches. The key advantage of NGS is that it is highly multiplexed to cover many genes in one test format, including genes for which no commercial molecular test exists. One of the key challenges to implementing NGS for this application is demonstrating the clinical value of such test formats to the medical community. BCC Research believes that this challenge is being aggressively addressed by a number of leading institutions in this field, as well as by several high-profile initiatives.
REPRODUCTIVE HEALTH APPLICATIONS
The main applications of NGS diagnostics in reproductive health are prenatal, newborn, and preimplantation screening. The first NGS test for prenatal screening was introduced by Sequenom in October 2011, and by mid-2017, more than 13 companies were marketing such tests. The key to this market opportunity was the development of technology that could detect and genetically analyze very low concentrations of fetal DNA in the peripheral circulation of pregnant women. The fetal DNA had to be accurately analyzed for chromosomal abnormalities that may lead to birth defects. Once this hurdle was overcome, the clinical benefit was rapidly recognized by the medical community.
Since launch of the initial tests, providers have sought to expand their testing menus to detect additional chromosomal abnormalities. This is a key strategy to getting higher market share in the at-risk population. The main clinical benefits of using NGS-based tests is a reduction in the number of invasive procedures and earlier diagnosis of fetal aneuploidy. Currently, NGS testing is mostly used to screen women at high risk of carrying babies with chromosomal abnormalities. A key issue that will impact future market potential is whether NGS-based screening will be adopted by the average-risk patient population. For that to happen, insurance providers and other payers will need to recognize a clear clinical benefit to screening this segment.
For newborn screening applications, NGS offers much promise as the technology continues down the cost curve. Newborn screening entails sequencing a baby’s genome at birth and keeping that information as a reference sequence throughout his or her life. The key driving force for this application is the decline in costs of sequencing and associated informatics, because mass screening is heavily dependent on costs. It is expected that sequencing costs will continue to decline; thus the future looks encouraging for this market application. The key hurdle that must be overcome for commercial success is demonstration of cost versus benefit and subsequent coverage of a screening test by insurance providers.
The first step is to show that NGS can duplicate the results of conventional newborn genetic tests, for which the current total cost is approximately $2,000. The next step is to demonstrate that NGS tests can provide more relevant clinical genetic information than conventional methods at a price point that is comparable or lower. If this can be done over the next few years, it is likely that insurance companies will come on board and a viable market will form.
Preimplantation screening using NGS is an emerging application. A key benefit is the potential to increase pregnancy success rates of in vitro fertilization. The main technological hurdle is being able to offer these tests at an attractive price point versus microarray methods and thereby increase the penetration of NGS. The tests do not rely on insurance coverage because they are offered with the option of cash payment. This feature has attracted the interest of a significant number of companies.
MICROBIOLOGY & INFECTIOUS DISEASE APPLICATIONS
The main applications of NGS diagnostics in microbiology and infectious diseases are 1) rapid pathogen detection and characterization, and 2) human immunodeficiency virus (HIV) tropism. Sequencing is playing an increasingly important role in the first market segment. The use of sequencing to diagnose patients who are hospitalized with food-borne infections is viewed as a near-term point of entry for NGS diagnostics. A key driving force for this particular application is the recent use of NGS to successfully manage disease outbreaks caused by food-borne pathogens (eg, Escherichia coli).
BCC Research envisions that the initial application of NGS for management of food-borne infection will be in hospitalized patients, representing only a small subset of individuals who become sick due to these infections. A main obstacle to commercialization will be education of clinicians on the benefits of using NGS to quickly identify pathogen strains so that effective treatments can be implemented. It will be important for developers to market the new tests appropriately to critical-care physicians and to contrast the benefits of NGS versus polymerase chain reaction (PCR) methods.
The comparison between NGS and PCR is a general issue in infectious disease testing. PCR is increasingly used in infectious disease diagnostics because it provides speed, accuracy, ease of use, and multiplex capabilities. Sanger sequencing, which is also used in some applications, provides highly accurate results too. NGS must compete with both technologies to meaningfully penetrate the infectious disease market.
The second application in which NGS has shown near-term commercial promise is HIV tropism testing, for which Sanger sequencing is the gold standard. However, NGS provides a better detection limit, such that mutations at lower allele frequencies can be detected. These lower-frequency mutations may be important in predicting drug resistance. It is thought that frequencies as low as 1% may influence drug sensitivity, and the present detection limit of Sanger sequencing is 20% frequency.
COMPLEX DISORDERS APPLICATIONS
The main applications of NGS diagnostics in this area include immune system, metabolic and mitochondrial, cardiovascular, and neurological disorders. Complex disorders are usually multifactorial, meaning there are many factors that determine who acquires the disease. These factors can be genetic or non-genetic (eg, environmental). Genetic factors may play only a minor role in some diseases, limiting the impact of NGS as a diagnostic. On the other hand, for many diseases, knowledge of their etiology remains incomplete and genetics may well play an important role. Thus, it is likely that as new knowledge is gained regarding the genetic basis of these diseases, the need for NGS diagnostics will increase.
For immune system disorders, main applications include immune system profiling, human leukocyte antigen (HLA) typing, rheumatoid arthritis (RA), and multiple sclerosis (MS). In the field of immune system profiling, NGS is making progress through both commercial assays and research. In addition, the Human Immunology Project Consortium is collecting all data generated by immune profiling techniques and putting it into a central, publicly accessible repository. This will help to accelerate this field.
Sanger sequencing is the current standard for high-resolution HLA typing. However, due to an increasing number of HLA alleles in the international ImMunoGeneTics database, histocompatibility testing is becoming more complex, even with Sanger sequencing. NSG assays provide rapid, high-resolution typing at a competitive cost. RA and MS are phenotypically complex, with underlying causes that are not well understood. Research into the genetic underpinnings of RA and MS is ongoing, and NGS technologies are useful in this effort. As this research progresses, it is expected that new molecular diagnostics will be developed, some of which will use NGS formats.
Mitochondrial disorders are difficult to diagnose because they have complex genetic causes and a wide range of phenotypes. These features make these diseases well suited for NGS diagnostics. Several laboratories have launched NGS-based clinical tests for mitochondrial disorders. Targeted panels are designed for cases in which a diagnosis is narrowed down to a small subset of mitochondrial diseases on the basis of phenotype, but further definition from a molecular diagnosis is needed. For this market segment, single-gene tests do not provide enough information, whereas whole-exome sequencing is too costly and overkill.
Cardiovascular and neurological disorders comprise a strong potential future market for NGS diagnostics. NGS tools have been used to demonstrate that breakdown in the control of gene expression may help to initiate or progress some of these diseases. A market for NGS diagnostics will coalesce when clinical research can correlate genetic changes with the risk of disease onset or progression. For example, it is important to determine if a patient with dilated cardiomyopathy has mutations in up to 40 genes that are associated with the disorder. NGS-based multigene tests are important diagnostic tools in the management of these patients. As a result, dilated cardiomyopathy is a focus for NGS test providers.
INTEGRATION OF CLINICAL NGS IN HEALTHCARE
NGS is playing an important role in the mission to transform cancer care, in particular. Examples include use of NGS in liquid biopsy pan-cancer panels; efforts to make gene sequencing and analysis services more accessible to physicians; and the emphasis on noninvasive liquid biopsy formats for early cancer detection, therapy selection, and monitoring. A high level of corporate deal-making illustrates the dynamic nature of the NGS market and reflects the promise of NGS in clinical applications. It also highlights the urgent need for companies to meet their goals toward delivering better clinical products and services to the healthcare industry.
This article is based on the following market analysis report published by BCC Research: DNA Sequencing: Emerging Clinical Applications and Global Markets (BIO126C) by John Bergin.
Laurie L. Sullivan, ELS, is a Bosto-based writer and editor with 20 years of experience in medical communications. She is certified by the Board of Editors in the Life Sciences. She contributes regularly to the BCC Research blog focusing on Life Sciences.
John Bergin is the author of previous BCC Research biotechnology reports and has held business development, sales, and marketing positions with a Fortune 500 advanced materials company, as well as executive management positions with a nanotechnology and separations company. Mr. Bergin earned his BS in Chemistry, his MS in Biotechnology, and a Master of Business Administration.
Total Page Views: 6535